|
4/11/2023 0 Comments Homo and lumo organic chemistry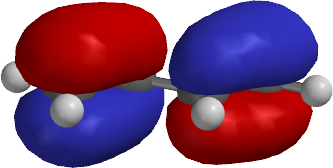
Molecules can begin to form bonds when they are able to share an electron. Why We Need the Frontier Molecular Orbital Theory It would be followed by HOMO +2 and so forth. The next highest occupied molecular orbital, for example, would be designated HOMO +1. HOMO stands for “highest occupied molecular orbital.” LUMO stands for “lowest unoccupied molecular orbital.” The “high” and “low” components of the description refer to the energies that are present.ĭifferent degrees of energy are present within these components. This is where the HOMO and LUMO interactions come into play. This means they tend to have the highest and lowest energies, whether they are occupied or unoccupied. These outer-edge orbitals, on the “frontier” of the molecule, are the ones that tend to be the most spatially delocalized. Why Is It Called the “Frontier” Molecular Orbital Theory?įrontier molecular theory looks at the orbitals which are at the outer edges of a molecule instead of all the orbitals that may exist. The occupied orbitals of one molecule and the unoccupied orbitals of the other molecule, with specificity to the HOMO and LUMO interactions, cause an attraction between the two molecules.īecause of these observations, the frontier molecular orbital theory can explain how the interactions of HUMO in one species are naturally attracted the LUMO of another species.The positive charges of one molecule with attract the negative charges of the other molecule.When there are occupied orbitals of different molecules, they will repel one another. 
Fukui made three primary observations for his theory as he watched two molecules interact with one another. The foundation of the theory is found by looking at the frontier orbitals, which are the HOMO and LUMO interactions. He would become the first Asian scientist to win a chemistry-based Nobel Prize. First published in the Journal of Chemical Physics by Kenichi Fukui in 1952, it is a theory of reactivity that would eventually help Fukui share a Nobel Prize in Chemistry for reaction mechanisms. Copyright © 2011 John Wiley & Sons, Ltd.Frontier molecular orbital theory is an application of the MO theory that describes the interactions of HOMO and LUMO interactions. These trends may provide insight into developing materials with specifically tuned HLGs and HOMO–LUMO levels for a variety of applications. The data presented not only elaborate on the HOMO–LUMO tuning of 9-fluorenone systems but also enable the consideration of 9-fluorenones as analogous models for HOMO–LUMO tuning in other more complex polyaromatic systems such as bifluorenylidenes. Spectroscopic evidence of substituent influence on the carbonyl suggests that substituents directly impact the HLG by influencing the availability of nonbonding electrons within the carbonyl, which impacts the probability of an nπ* transition. Increasing conjugation decreased the HLG, increased the HOMO energy level, but decreased the LUMO energy level. Increasing the electron-donating character of the substituents was observed to decrease the HLG and increase the energy levels of the HOMO and the LUMO, whereas an increase in the electron-withdrawing character produced the opposite results. Results from both methods were compared and correlated with the differences in molecular structure. Electrochemical and optical measurements were used to calculate the HOMO–LUMO levels and HOMO–LUMO bandgap (HLG) for each structure. Compounds with an incremental increase in conjugation were also examined. Trends were explored in a range of compounds, beginning with structures having highly electron-withdrawing substituents and progressing to structures having highly electron-donating substituents. A study of the specific effects and overall trends for the HOMO–LUMO tuning of a diverse series of 9-fluorenones by means of extended conjugation and substituent effects is described. Highest occupied molecular orbital–lowest unoccupied molecular orbital (HOMO–LUMO) tuning is an important consideration in the development of organic-based semiconducting materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
